Have you ever wondered about the mysterious world of covalent bonds? These bonds play a crucial role in chemistry, holding atoms together to form molecules. Let’s dive into the fascinating world of covalent bonds and explore one in particular: S2Cl4.
Covalent bonds are formed when two atoms share electrons to achieve a stable electron configuration. In the case of S2Cl4, sulfur forms bonds with chlorine atoms by sharing electrons. This results in a stable molecule with a unique structure.
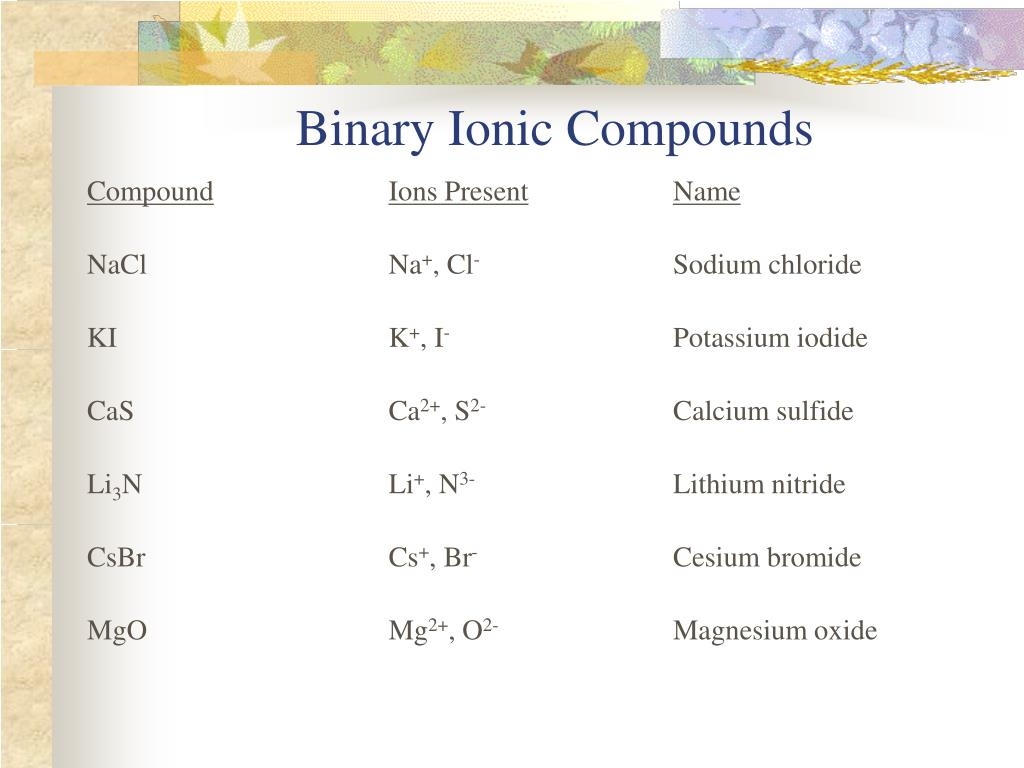
name the covalent bond: s2cl4
Name the Covalent Bond: S2Cl4
S2Cl4, also known as disulfur tetrachloride, is a chemical compound composed of two sulfur atoms and four chlorine atoms. The covalent bonds between sulfur and chlorine atoms give S2Cl4 its distinct properties and characteristics.
Disulfur tetrachloride is a colorless liquid with a pungent odor, commonly used in the synthesis of other sulfur compounds. Its molecular structure consists of a central sulfur atom bonded to two other sulfur atoms and four chlorine atoms, creating a tetrahedral shape.
When naming covalent compounds like S2Cl4, it is essential to understand the chemical composition and bonding arrangement. By studying the structure of molecules, scientists can predict their properties and behavior in various chemical reactions.
In conclusion, covalent bonds are vital in the world of chemistry, playing a significant role in the formation of molecules like S2Cl4. Understanding the structure and properties of covalent compounds enhances our knowledge of chemical reactions and their applications in various industries.

Unit B Matter And Chemical Change Notes naming Molecular Compounds PPTX

Unit B Matter And Chemical Change Notes naming Molecular Compounds PPTX
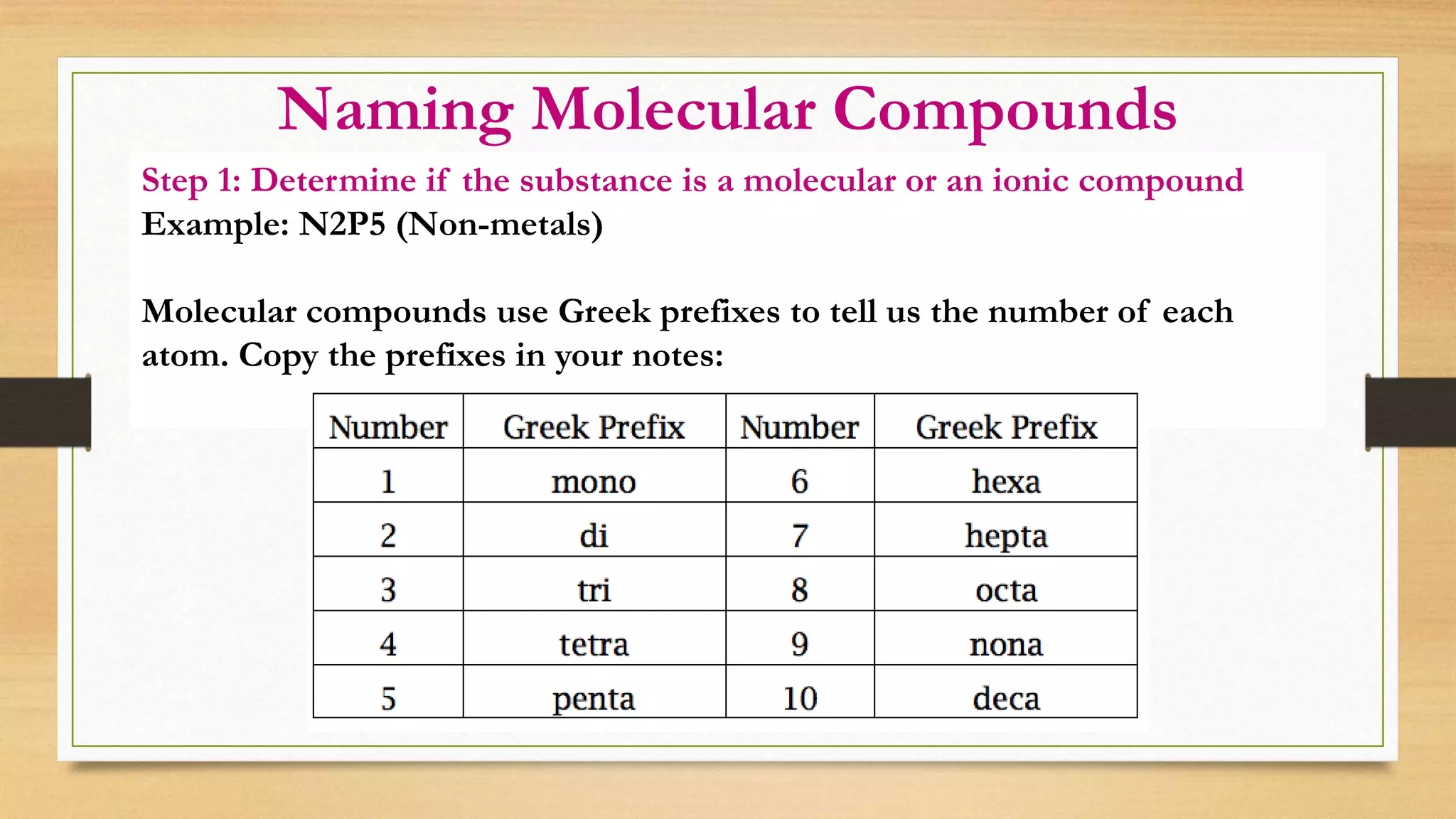
Unit B Matter And Chemical Change Notes naming Molecular Compounds PPTX

Unit B Matter And Chemical Change Notes naming Molecular Compounds PPTX
